- Blog
- Rapelay play rl don-t work
- Superpowered -17 walkthrough
- Gamecube xbox 360 rgh emulator
- Periodic table with molar masses and charges
- Download teamviewer quick support apk
- Change samsung folder icon color
- Vuze requiring root access to update
- Suprabhatam ms subbulakshmi original mp3 download
- T value degrees of freedom calculator
- Bongiovi dps for windows 10 crack
- Ufc 3 pc download crack
- November 2 starfall calendar
- Herchey script font free download
- Adobe flash player 9 or higher download
- East west symphonic orchestra vs hollywood
- Aesthetic youtube banner 2560x1440
- Shadowrun 5e form fillable character sheet
- Inuyasha season 3 torrent
- Work schedule monthly calendar templates free
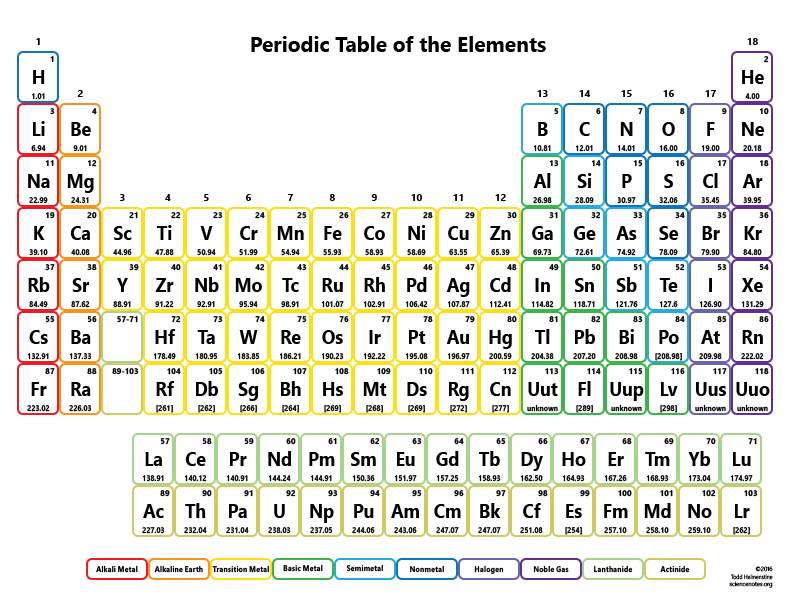
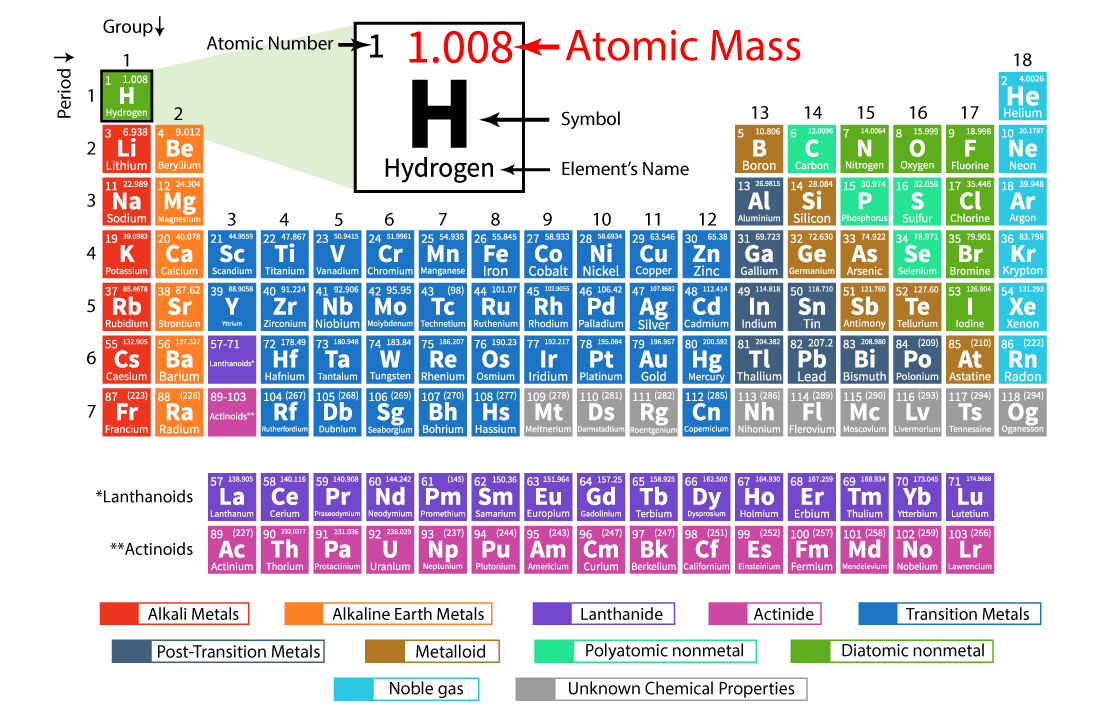
Atoms of the same element that have a different number of neutrons are called isotopes. Let us now learn the difference between atomic number and atomic mass of elements.īefore going into atomic mass, it is essential to learn about isotopes. This was the atomic mass of the first 30 elements. Given below is the atomic mass of elements list. Let us take a look at what is the atomic mass of elements from 1 to 30. In the same group, a periodic repetition of properties can be seen in the elements with increasing mass. Similarly, the elements in Group 8A are unreactive gaseous at room temperature. For example, the elements in Group 1A are mostly soft metals that are highly reactive with water. Elements that have similar qualities fall into the same column or group. When observing the periodic table, one can see that elements are arranged on the basis of increasing atomic numbers. The number of electrons in an element considerably determines the chemical behaviour of the element. The atomic number is important because the number of protons determines the number of electrons that surround the nucleus. Atoms of all elements have neutrons in the nucleus except hydrogen. Neutrons are uncharged subatomic particles which are stable when bound in an atomic nucleus. Elements are identified based on the number of protons in the nucleus regardless of the number of neutrons present. The atomic number gives a number of how many protons are inside the nucleus of the atom. The overall atomic masses that are given in periodic tables like the one for hydrogen are determined for the naturally occurring isotopes of each element, weighted by the weight of those particular isotopes on earth. Every isotope's contribution to the normal is controlled by how huge a fraction of the example it makes up. The general atomic mass is the normal of the atomic masses of the apparent multitude of various isotopes in an example. Since an element's isotopes have distinctive atomic masses, researchers may likewise decide the general atomic mass-once in a while called the atomic weight-for an element. The atomic mass number is usually rounded off to the nearest whole number. For example, a normal carbon atom with six neutrons and six protons is denoted as carbon-12. The atomic mass of a solitary atom is its absolute mass and is regularly expressed in atomic mass units or amu. Since we have seen the definition of atomic mass let us discuss it in detail. In this article, we will learn about the following things: the atomic mass of elements in detail, what is the atomic mass of all elements, and what is the atomic number and atomic mass of elements. An interesting point to note is that it is also referred to as atomic weight. The atomic mass is simply defined as the weighted average of all of the isotopes of an element, in which the mass of each isotope is multiplied by the abundance of that particular isotope. Therefore, there are various non-equivalent definitions of atomic radius.The atomic mass in Chemistry is the average mass of the atoms of an element measured in atomic mass units (amu). However, this assumes the atom to exhibit a spherical shape, which is only obeyed for atoms in vacuum or free space.
The atomic radius of a chemical element is a measure of the distance out to which the electron cloud extends from the nucleus. It must be noted, atoms lack a well-defined outer boundary.
The atomic radius of Vanadium atom is 153pm (covalent radius). Note that, each element may contain more isotopes, therefore this resulting atomic mass is calculated from naturally-occuring isotopes and their abundance. The atomic mass is carried by the atomic nucleus, which occupies only about 10 -12 of the total volume of the atom or less, but it contains all the positive charge and at least 99.95% of the total mass of the atom. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. Mass numbers of typical isotopes of Vanadium are 51. Isotopes are nuclides that have the same atomic number and are therefore the same element, but differ in the number of neutrons. The difference between the neutron number and the atomic number is known as the neutron excess: D = N – Z = A – 2Z.įor stable elements, there is usually a variety of stable isotopes. Neutron number plus atomic number equals atomic mass number: N+Z=A. The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol N. The total electrical charge of the nucleus is therefore +Ze, where e (elementary charge) equals to 1,602 x 10 -19 coulombs. Total number of protons in the nucleus is called the atomic number of the atom and is given the symbol Z.

Vanadium is a chemical element with atomic number 23 which means there are 23 protons in its nucleus. Atomic Number – Protons, Electrons and Neutrons in Vanadium
- Blog
- Rapelay play rl don-t work
- Superpowered -17 walkthrough
- Gamecube xbox 360 rgh emulator
- Periodic table with molar masses and charges
- Download teamviewer quick support apk
- Change samsung folder icon color
- Vuze requiring root access to update
- Suprabhatam ms subbulakshmi original mp3 download
- T value degrees of freedom calculator
- Bongiovi dps for windows 10 crack
- Ufc 3 pc download crack
- November 2 starfall calendar
- Herchey script font free download
- Adobe flash player 9 or higher download
- East west symphonic orchestra vs hollywood
- Aesthetic youtube banner 2560x1440
- Shadowrun 5e form fillable character sheet
- Inuyasha season 3 torrent
- Work schedule monthly calendar templates free
